Provein Safe 14G IV Cannula
$62.99
$90.08
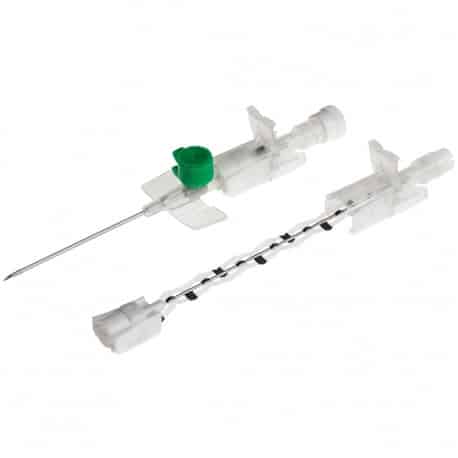
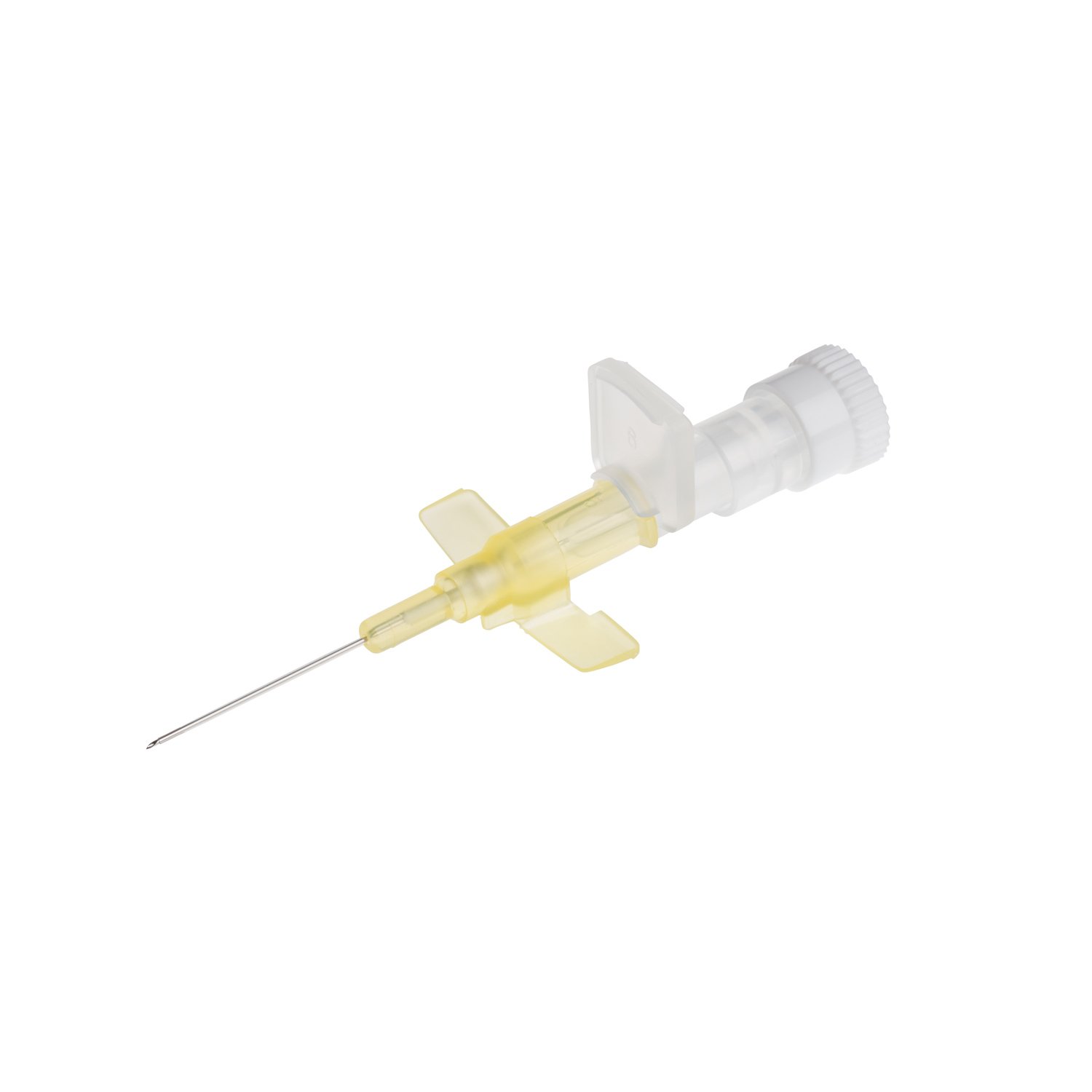
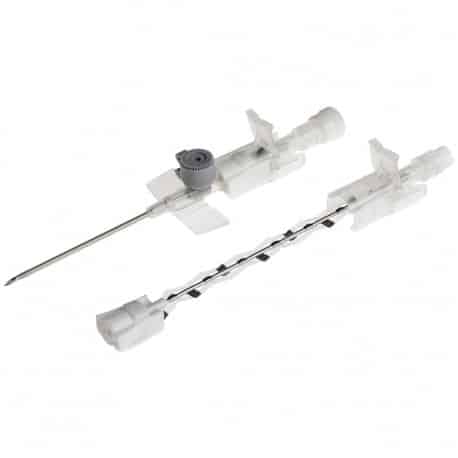
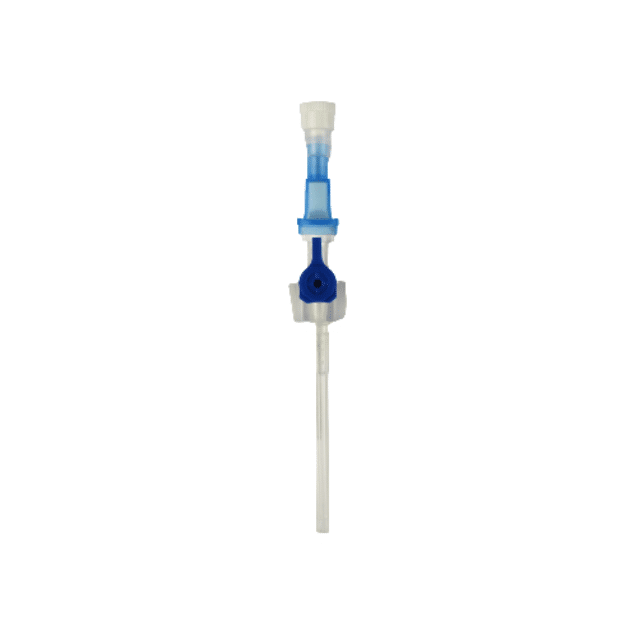
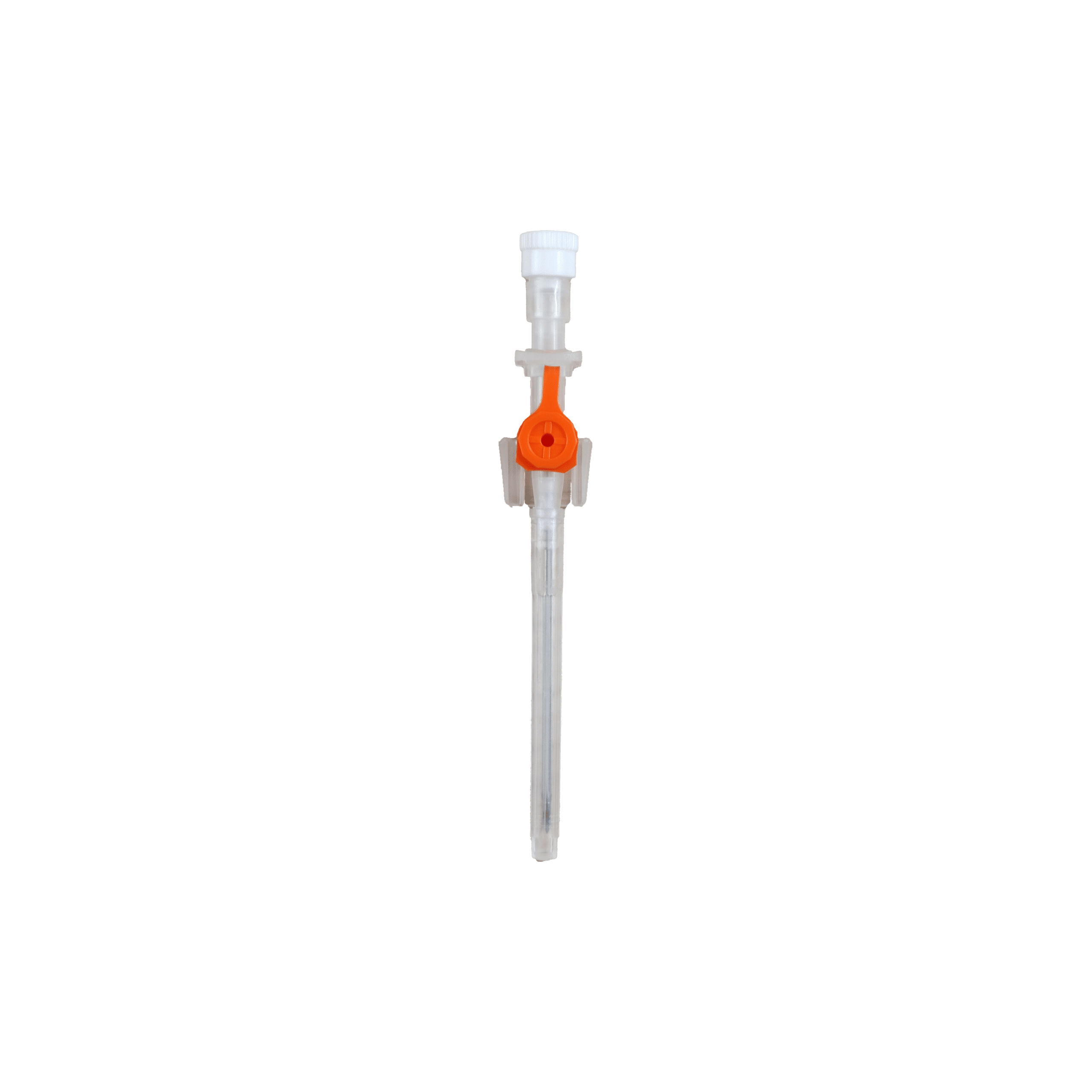
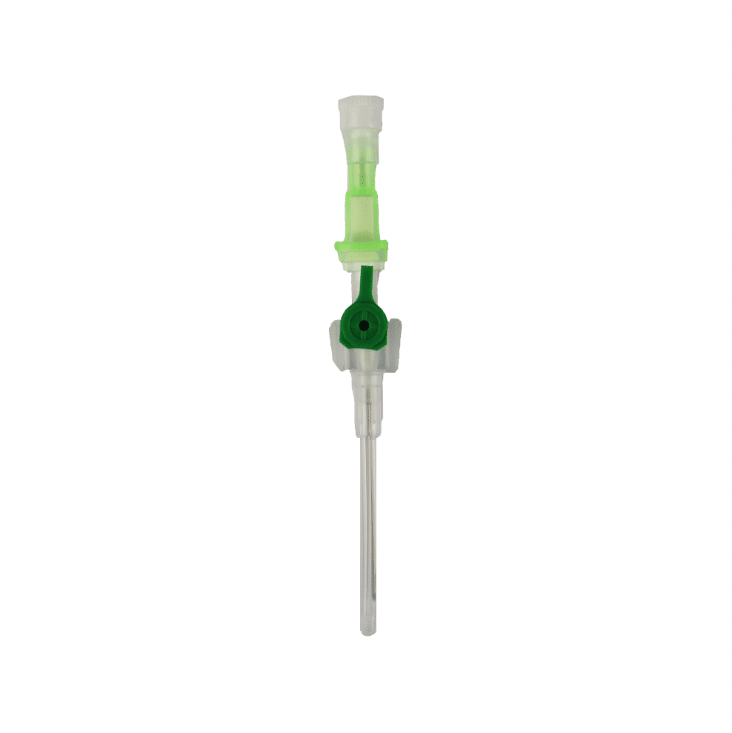
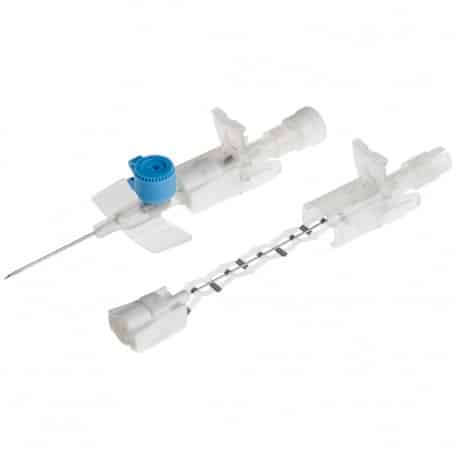
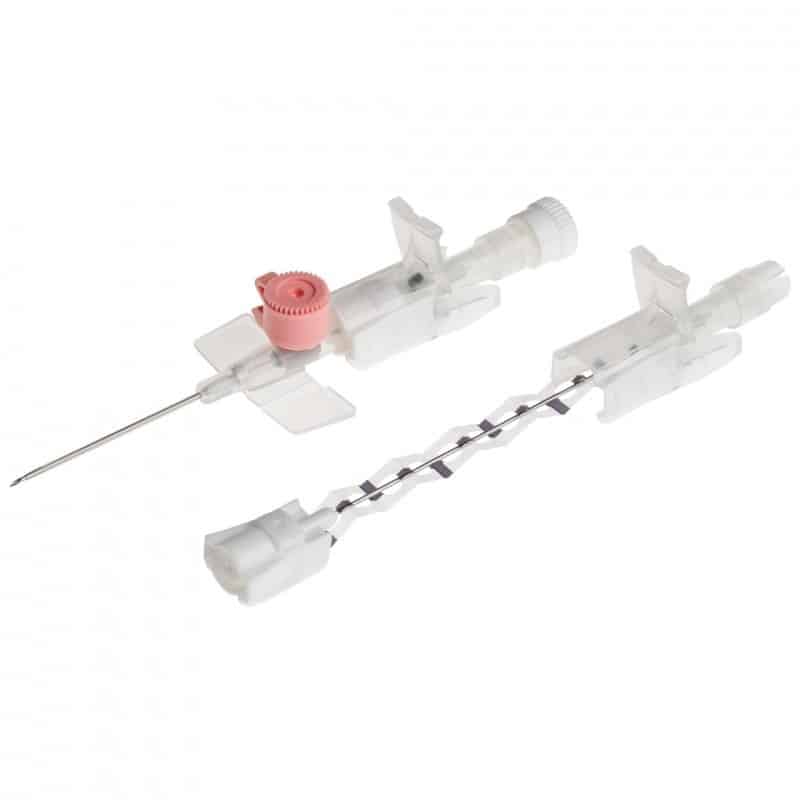
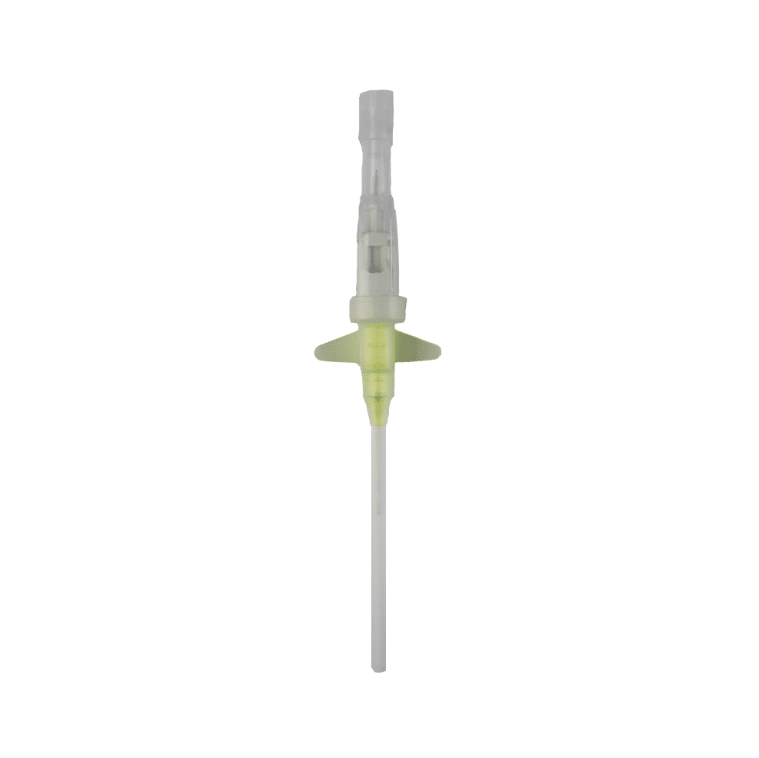
The Provein Safe 14G IV Cannula is the largest gauge peripheral venous access device designed for critical emergency scenarios requiring maximum flow rates. With an ultra-high flow capacity of approximately 250 ml/min, this orange-coded cannula is the standard choice for trauma resuscitation, rapid blood transfusions, and emergency fluid replacement. Manufactured by LARS Medicare – a family-run medical device company with over 20 years of ISO 13485-certified manufacturing excellence – the Provein Safe combines clinical performance with integrated safety features to protect healthcare professionals from needlestick injuries. Key Clinical Applications Emergency trauma resuscitation and hemorrhagic shock management Massive blood transfusions and rapid blood product delivery Emergency surgery preparation requiring rapid fluid loading Critical care high-volume fluid management in ICU settings Major burn injuries requiring rapid resuscitation protocols Technical Specifications Specification Details Gauge Size 14G (2.0mm outer diameter) Color Coding Orange (international standard) Catheter Material PTFE (Polytetrafluoroethylene) – biocompatible, ultra-smooth Needle Material Medical-grade stainless steel, precision-ground bevel Catheter Length 45mm (standard peripheral insertion) Maximum Flow Rate ~250 ml/min (highest peripheral cannula flow) Safety Mechanism Integrated one-handed needle safety device Design Features Flexible wings, injection port for secondary access Sterility ETO sterilized, individually sealed packaging Latex Status Latex-free (hypoallergenic) Design Features & Clinical Benefits PTFE Catheter Technology The Provein Safe utilises PTFE catheter material – the gold standard for biocompatibility and ultra-smooth insertion. PTFE’s low-friction coefficient reduces vascular trauma during advancement and minimizes thrombophlebitis risk during extended use. Integrated Safety Mechanism One-handed needle safety activation shields the sharp needle immediately after catheter insertion, reducing needlestick injury risk by over 80% compared to conventional cannulas. Critical in high-stress emergency scenarios. Clear Flashback Chamber Transparent flashback chamber provides immediate visual confirmation of successful venipuncture, reducing vessel trauma from repeated insertion attempts. Saves precious seconds in life-threatening situations. Flexible Wings & Injection Port Bilateral stabilization wings provide ergonomic handling during insertion and secure fixation with medical tape. The integrated injection port allows secondary medication administration without interrupting primary fluid delivery. When to Choose 14G vs. Other Gauges 14G is optimal when: Maximum flow rate is clinically essential (trauma, hemorrhagic shock, massive transfusion protocols) Adult patients with large-caliber peripheral veins (antecubital access) Emergency department or trauma bay scenarios Surgical procedures requiring rapid intraoperative fluid shifts Consider smaller gauges when: Flow rates of 200 ml/min (16G) or 130 ml/min (18G) are clinically sufficient Patient has moderate veins where 14G may cause excessive trauma Patient comfort prioritized in non-emergency scenarios Manufactured by LARS Medicare: Family-Trusted Quality Since 2005 LARS Medicare Pvt. Ltd. is a family-run medical device manufacturer with over 20 years of proven excellence in IV cannula production. Founded by Sanjay Goel in 2005 and now managed across two generations of the Goel family, LARS Medicare embodies consistent quality, long-term partnership, and unwavering commitment to healthcare professionals worldwide. Quality Credentials: ISO 13485:2003 Certified – Medical device quality management CE Marked – Recognised for Great Britain market MHRA Registered – UK compliant In-House Testing Laboratory – Three-level quality validation Computer-Controlled EO Sterilization Exported to over 70 countries across Asia Pacific, Latin America, Europe, and beyond. Regulatory Compliance & Safety CE Marked: EU Medical Device Regulation compliant; recognized in Great Britain MHRA Registered: Approved for distribution in England, Wales, Scotland UK Responsible Person: Reflex Medical Limited, England Single-Use Device: Sterile, non-pyrogenic, one-time use only Latex-Free: Suitable for latex-allergic patients Biocompatibility: ISO 10993 compliant Ordering Information Packaging: 50 units per box, individually sterile packed Bulk Ordering: Available – contact us for information. Stock: Held in UK for rapid delivery
Iv Cannulas